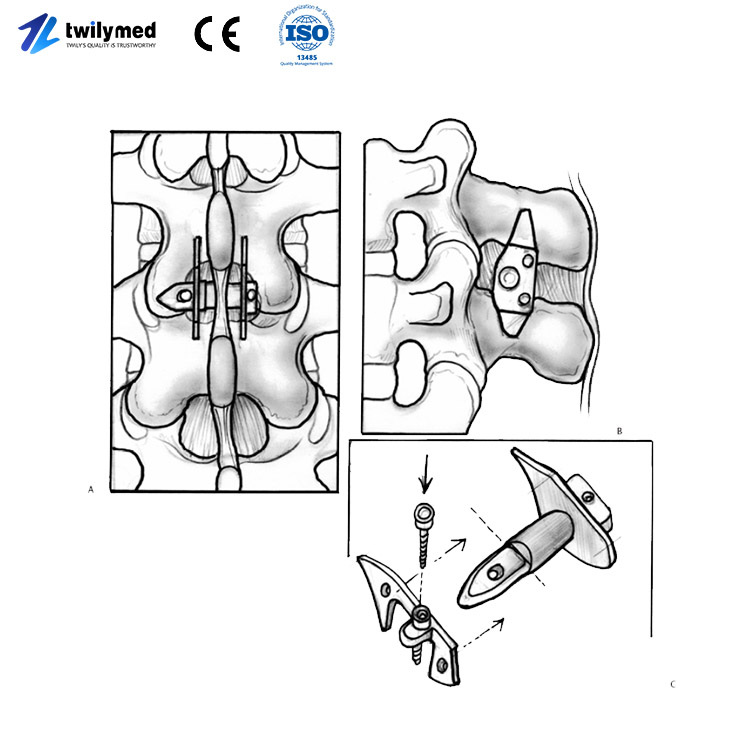
Spinal Interspinous Spacer.

These devices are primarily used to treat lumbar spinal stenosis (LSS) and related degenerative diseases. Their core design concept involves implanting the spacer between the spinous processes of two adjacent vertebrae through minimally invasive surgery, acting as a “pad.”

They expand the spinal canal space: limiting spinal extension, thereby increasing the area of the spinal canal and neural foramina, reducing compression on nerve roots.

They maintain dynamic stability: while reducing compression, they preserve a certain degree of spinal mobility, a technique known as “dynamic stabilization.”
They relieve pain: helping to alleviate symptoms such as lower back pain, hip pain, and leg numbness and tingling (intermittent claudication) that worsen with standing or walking.
Common Products and Specifications
Based on visual search results, these products are typically made of titanium alloys (such as Ti-6Al-4V) or PEEK (polyetheretherketone) materials, offering good biocompatibility. Common sizes: They are usually available in a variety of sizes to accommodate different patient anatomy, such as 6mm, 8mm, 10mm, 12mm, 14mm, 16mm, etc.
















Reviews
There are no reviews yet.